Coming of Age in the Milky Way (38 page)
Read Coming of Age in the Milky Way Online
Authors: Timothy Ferris
Tags: #Science, #Philosophy, #Space and time, #Cosmology, #Science - History, #Astronomy, #Metaphysics, #History

T
HE
E
VOLUTION OF
A
TOMS AND
S
TARS
At quite uncertain times and places,
The atoms left their heavenly path,
And by fortuitous embraces,
Engendered all that being hath.—James Clerk Maxwell
For I have already at times been a boy and a girl, and a bush and a bird and a mute fish in the salty waves.
—Empedocles
B
y the dawn of the twentieth century it was becoming evident that some sort of “atomic” energy must be responsible for powering the sun and the other stars. As early as 1898, only two years after Becquerel’s discovery of radioactivity, the American geologist Thomas Chrowder Chamberlin was speculating that atoms were “complex organizations and seats of enormous energies” and that “the extraordinary conditions which reside in the center of the sun may … set free a portion of this energy.”
1
But no one could say what this mechanism might be, or just how
it might operate, until a great deal more was understood about both atoms and stars. The effort to garner such an understanding involved a growing level of collaboration between astronomers and nuclear physicists. Their work was to lead, not only to a resolution of the stellar energy question, but to the discovery of a golden braid of cosmic evolution intertwining atomic and stellar history.
The key to understanding stellar energy was, as Chamberlin foresaw, to discern the structure of the atom. That there
was
an internal structure to the atom could be intimated from several lines of research, among them the study of radioactivity: For atoms to emit particles, as they were found to do in the laboratories of Becquerel and the Curies, and for these emissions to change them from one element to another, as Rutherford and the English chemist Frederick Soddy had established, atoms must be more than the simple, indivisible units that their name (from the Greek for “cannot be cut”) implied. But atomic physics still had a long way to go in comprehending that structure. Of the three principal constituents of the atom—the proton, neutron, and electron—only the electron had as yet been identified (by J. J. Thomson, in the waning years of the nineteenth century). Nobody spoke of “nuclear” energy, for the existence of the atomic nucleus itself had not been established, much less that of its constituent particles the proton and the neutron, which were to be identified, respectively, by Thomson in 1913 and James Chadwick in 1932.
Rutherford, Hans Geiger, and Ernest Marsden ranked among the Strabos and Ptolemies of atomic cartography. In Manchester from 1909 through 1911 they probed the atom by launching streams of subatomic “alpha particles”—helium nuclei—at thin foils made of gold, silver, tin, and other metals. Most of the alpha particles flew through the foil, but, to the experimenters’ astonishment, a few bounced right back. Rutherford thought long and hard about this strange result; it was, he remarked, as startling as if a bullet were to bounce off a sheet of tissue paper. Finally, at a Sunday dinner at his house in 1911, he announced to a few friends that he had hit on an explanation—that most of the mass of each atom resides in a tiny, massive nucleus. By measuring the back-scattering rates obtained from foils comprised of various elements, Rutherford could calculate both the charge and the maximum diameter of the atomic nuclei in the target. Here, then, was an atomic explanation
of the weights of the elements: Heavy elements are heavier than light elements because the nuclei of their atoms are more massive.
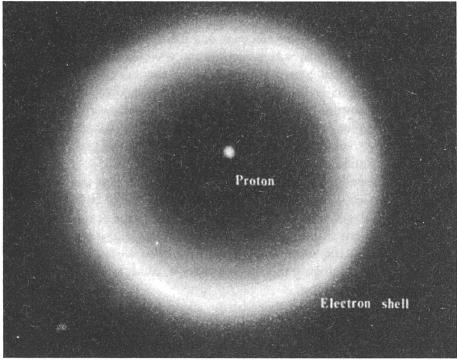
An atom of simple hydrogen consists of a single proton (its nucleus) surrounded by a shell containing one electron. Atoms of heavier elements have more protons, as well as neutrons, in the nucleus, and additional electrons in the shells. (Not to scale: Were the proton the size of a grain of sand, the shell would be larger than a football field.)
The realm of the electrons was then explored by the Danish physicist Niels Bohr, who established that electrons inhabit discrete orbits, or shells, surrounding the nucleus. (For a time Bohr thought of the atom as a miniature solar system, though this analysis soon proved inadequate; the atom is ruled not by Newtonian but by quantum mechanics.) Among its many other felicities, the Bohr model laid bare the physical basis of spectroscopy: The number of electrons in a given atom is determined by the electrical charge of the nucleus, which in turn is due to the number of protons in the nucleus, which is the key to the atom’s chemical identity. When
an electron falls from an outer to an inner orbit it emits a photon. The wavelength of that photon is determined by the particular orbits between which the electron has made the transition. And
that
is why a spectrum, which records the wavelengths of photons, reveals the chemical elements that make up the star or other object the spectroscopist is studying. In the words of Max Planck, the founder of quantum physics, the Bohr model of the atom provided “the long-sought key to the entrance gate into the wonderland of spectroscopy, which since the discovery of spectral analysis had obstinately defied all efforts to breach it.”
2
But, marvelous though it might be to realize that the spectra evinced the leaps and tumbles of electrons in their Bohr orbits, nobody could yet read the spectra of stars for significant clues to what made them shine. In the absence of a compelling theory, the field was left to the taxonomists—to those who went on doggedly recording and cataloging the spectra of stars, though they knew not where they were going.
At Harvard College Observatory, a leader in the dull but promising business of stellar taxonomy, photographic plates that revealed the color and spectra of tens of thousands of stars were stacked in front of “computers”—spinsters, most of them, employed as staff members at a university where their sex barred them from attending classes or earning a degree. (Henrietta Leavitt, the pioneer researcher of the Cepheid variable stars that were to prove so useful to Shapley and Hubble, was a Harvard computer.) The computers were charged with examining the plates and entering the data in neat, Victorian script for compilation in tomes like the
Henry Draper Catalog
, named in honor of the astrophotographer and physician who had made the first photograph of the spectrum of a star. Like prisoners marking off the days on their cell walls, they tallied their progress in totals of stars cataloged; Antonia Maury, Draper’s niece, reckoned that she had indexed the spectra of over five hundred thousand stars. Theirs was authentically Baconian work, of the sort Newton and Darwin claimed to practice but seldom did, and the ladies took pride in it; as the Harvard computer Annie Jump Cannon affirmed, “Every fact is a valuable factor in the mighty whole.”
3
It was Cannon who, in 1915, first began to discern the shape of that whole, when she found that most stars belonged to one of about a half-dozen distinct spectral classes. Her classification system, now ubiquitous in stellar astronomy, arranges the spectra of
the stars by color, from the blue-white O stars through yellow G stars like the sun to the red M stars.
*
Here was a sign of simplicity beneath the astonishing variety of the stars.
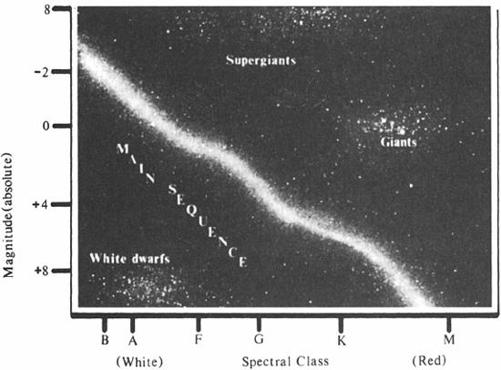
A still deeper order was soon disclosed, when in 1911 the Danish engineer and self-taught astronomer Ejnar Hertzsprung analyzed Cannon’s and Maury’s data for stars in two clusters, the Hyades and the Pleiades. Clusters like these are intuitively recognizable as genuine assemblies of stars and not merely chance alignments; even an inexperienced observer will start with recognition when sweeping a telescope across the Pleiades, its ice-blue stars tangled in gossamer webs of diamond dust, or the Hyades, whose stars range in color from bone-white to Roman gold. Since all the stars in a given cluster may thus be assumed to lie at about the same distance from the earth, any observed differences in their apparent magnitudes can be ascribed, not to their differing distances, but to actual differences in their absolute magnitudes. Hertzsprung took advantage of this situation, treating the clusters as laboratory samples wherein he could look for a relationship between the colors and intrinsic brightnesses of stars. He found just such a relationship: Most of the stars in each cluster fell along two gently curved lines. This, in sapling form, was the first intimation of a tree of stars that has since been designated the Hertzsprung-Russell diagram.
The applicability of Hertzsprung’s method soon broadened to include noncluster stars as well. In 1914, Walter Adams and Arnold Kohlschutter at Mount Wilson found that the relative intensities of lines in stellar spectra suggested their absolute magnitudes. Hereafter, whenever the distance to a single star of a given variety—a class B giant, say, or a class K dwarf—was measured by the parallax method, then the distances of all other stars that displayed comparable spectra could be estimated as well. This meant that Hertzsprung’s approach of graphing the absolute magnitude of stars against their colors could be applied to field stars as well as to the relatively few stars found in clusters.
Henry Norris Russell, a Princeton astrophysicist with an encyclopedic
command of his field, promptly set to work doing just that. Without even knowing of Hertzsprung’s work, Russell plotted the absolute magnitudes of a few hundred field stars against their colors, and found that most lie along a narrow, slanting zone—the trunk of the tree of stars.
The tree has been growing ever since, and today it is embedded in the consciousness of every stellar astronomer in the world. Its trunk is the “main sequence,” a gently curving S along which lie 80 to 90 percent of all visible stars. The sun, a typical yellow star, resides on the main sequence a little less than halfway up the trunk. A slender branch departs from the trunk and makes its way upward and to the right, where it blossoms into a bouquet of brighter, redder stars—the red giants. Below and to the left sits a humus pile of dim, blue to white stars—the dwarfs.
The Hertzsprung-Russell diagram provided astronomers with a frozen record of evolution, the astrophysical equivalent of the fossil record geologists study in rock strata. Presumably, stars somehow evolve, spending most of their time on the main sequence (most stars today, in the snapshot of time given us to observe, are found there) but beginning and ending their careers somewhere else, among the branches or in the humus pile. One could not, of course, wait to see this happen; the lifetimes of even short-lived stars are measured in millions of years. To find the answers required understanding the physics of how stars work.
Progress on the physics side, meanwhile, was blocked by a seemingly insurmountable barrier. Literally so: The agency responsible was known as the Coulomb barrier, and for a time it stymied the efforts of theoretical physicists to comprehend how nuclear fusion might produce energy in stars.
The line of reasoning that led to the barrier was impeccable. Stars are made mostly of hydrogen. (This is evident from studying their spectra.) The nucleus of the hydrogen atom consists of but a single proton, and the proton contains nearly all the mass in the atom. (This we know from Rutherford’s experiments.) Therefore the proton must also contain nearly all of a hydrogen atom’s latent energy. (Recall that mass equals energy:
E = mc
2
). In the heat of a star, the protons are flung about at high velocities—heat
means
that the particles involved are moving fast—and, as there are plenty of protons milling about in close quarters at the dense core of a star, they must collide quite a lot. In short, the energy of the sun
and stars could reasonably be assumed to involve the interactions of protons. This was the basis of Eddington’s surmise that the stellar power source could “scarcely be other than the subatomic energy which, it is known, exists abundantly in all matter.”
4