U.S. Army Special Forces Guide to Unconventional Warfare (4 page)
Read U.S. Army Special Forces Guide to Unconventional Warfare Online
Authors: Department of the Army

a. Description.
- This item consists of a small pile of potassium permanganate crystals which are ignited by the chemical action of glycerin on the crystals. It can be used to ignite all the incendiaries listed in chapter 4 except Thermite (0307). It may be used directly as an incendiary on readily flammable material, such as rags, dry paper; dry hay, or in the combustible vapor above liquid fuels.
- Ignition is accomplished by causing a few drops of glycerin to contact the potassium permanganate crystals. A hotter flame is produced when powdered magnesium or powdered aluminum is mixed with the the potassium permanganate crystals.
- Ignition time, after addition of the glycerin, increases as temperature decreases. This igniter is not reliable below 50° F.
b. Material and Equipment.
Potassium permanganate crystals (no coarser than granulated sugar).
Glycerin.
One small container with tight-fitting lid for the glycerin.
One larger container with tight-fitting lid for the potassium permanganate crystals.
Powdered magnesium or powdered aluminum (no coarser than granulated sugar).
Preparation.
- Put some glycerin in the small containers and cap tightly.
- Fill the larger container with potassium permanganate crystals and cap tightly.
- If powdered magnesium or powdered aluminum is available, mix 85 parts potassium permanganate crystals and 15 parts powdered magnesium or powdered aluminum and store this mixture in the large bottle.
- Keep these containers tightly sealed and the material in the containers will remain effective for a long period of time.
d. Application.
Pour out a quantity of the potassium permanganate crystals (with or without powdered aluminum or powdered magnesium), in a single pile on the incendiary. Manual ignition is accomplished by causing a few drops of glycerin from a medicine dropper to come in contact with the potassium permanganate crystals. Keep hands and clothing clear of the igniter; ignition may take place almost instantly with addition of the glycerin. This igniter is convenient for use with specific delay mechanisms found in chapter 5.
a. Description.
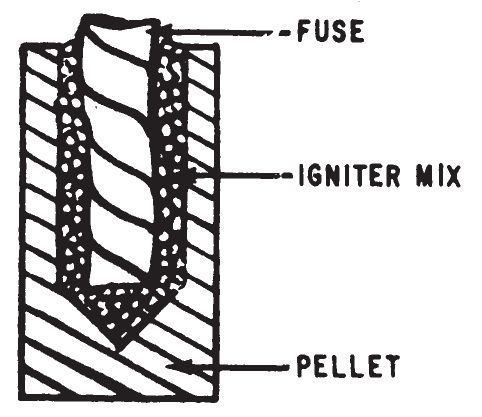
- This igniter consists of finely powdered aluminum, sulfur and starch which have been thoroughly mixed and shaped into hardened cylindrical pellets. It can be used to ignite all the incendiaries listed in chapter 4. It is an excellent igniter for Thermite (0307). It may be used directly as an incendiary on readily flammable material such as rags, dry paper, dry hay, or in the combustible vapor above liquid fuels.
- The igniter can be initiated by Fuse Cord (0101) or Improvised String Fuse (0102). A hole is made in one pellet to receive a fuse and a small quantity of another more easily started igniter mixture. A number of unmodified pellets are attached to the first pellet to increase the quantity of heat after combustion occurs.
b. Material and Equipment.
Finely powdered aluminum (no coarser than cake flour).
Finely powdered sulfur (no coarser than cake flour).
Finely powdered starch (no coarser than cake flour) .
Water.
Cylindrical tube about 4 inches long and ¾ inch inside diameter made of metal, wood, glass or plastic.
Rod which fits into the above tube.
Rod about â
inch in diameter (should be about one-half the inside diameter of the 4-inch long tube).
Mixing bowl.
Tablespoon.
Teaspoon.
Stove or hot plate.
Knife.
Measuring container.
c. Preparation.
- Place six tablespoons of aluminum powder in a mixing bowl then add 15 tablespoons of powdered sulfur.
- Mix the two powders gently with the spoon for a few minutes until no unmixed particles of sulfur are visible.
- In a separate pot add two teaspoons of laundry starch to about 6 ounces of water and boil gently for a few minutes. Stir until the starch is dissolved and allow the solution to cool to room temperature.
- When cool, take about one-half of the starch solution and add it to the mixture of aluminum and sulfur powder.
- Mix with a spoon until the whole mass is a smooth, evenly mixed, putty-like paste.
- Fill the cylindrical tube with this paste, place one end of this tube on a hard surface and tamp the paste with the â
inch diameter rod to squeeze out the air bubbles and consolidate the paste.
- Push the paste out of the tube with the larger rod, which just fits the tube, so that it forms a cylinder, then cut the damp cylinder into 1½ inch lengths using the knife.
- Dry these pieces at 90° F. for at least 24 hours before using. The drying time can be reduced by using a drying oven at a maximum temperature of 150° F.
- Form a hole at least ½ inch in diameter approximately half-way into one end of an igniter pellet.
- Put one of the following igniters into the cavity to roughly one-half its depth:
Sugar-Chlorate (0201)
SugarâSodium Peroxide (0203)
Aluminum PowderâSodium Peroxide (0204)
Silver NitrateâMagnesium Powder (0208)
- Insert a length of fuse into the hole so that it makes contact with the igniter mix. Fill the remainder of the hole with igniter mix and tamp down to hold the fuse firmly.
- Tape the fuse cord in place to prevent it from working loose and falling out.
- Tape two or more pellets without holes to the one with the fuse.
- Store all the pellets in a dry, closed container until required for use.
d. Application.
- For ignition of thermite, a cluster of at least three pellets should be used. Bury the cluster of igniter pellets just below the surface of the thermite, with the fuse extending for ignition by a match flame. Large quantities of thermite may require a cluster of more than three pellets for satisfactory ignition.
- For use as an igniter of a solid incendiary, place a cluster of pellets on top of the incendiary.
- When used to ignite flammable liquids, wrap a cluster of igniter pellets in a nonabsorbent material and suspend it inside the container near the open top. The container must remain open for easy ignition and combustion of the flammable liquid.
- To minimize the hazard of premature ignition of flammable liquid vapors, allow at least two feet of fuse length to extend from the top edge of an open container of flammable liquid before lighting the fuse.
a. Description.
- This item consists of a mixture of silver nitrate crystals and magnesium powder. It can be used to ignite all the incendiaries listed in chapter 4 except Thermite (0307). It may be used directly as an incendiary on readily flammable material such as rags, dry paper, dry hay, or in the combustible vapor above liquid fuels.
- The igniter can be initiated by Fuse Cord (0101), Improvised String Fuse (0102), Concentrated Sulfuric Acid (0103), or Water (0104).
Caution:
This mixture is unstable and may ignite at high humidity or when wet slightly by drops of water, perspiration, etc.
b. Material and Equipment.
Silver nitrate crystals (no coarser than granulated sugar).
Magnesium powder or filings (no coarser than granulated sugar).
Spoon.
Container with tight-fitting lid.
c. Preparation.
- Using a clean, dry spoon, place magnesium powder or filings into the dry mixing container to one-quarter container volume. If magnesium filings are used, they should be free of grease.
- Wipe the spoon with a clean, dry cloth, then add an equal quantity of silver nitrate crystals to the dry mixing container. Tighten the lid on the silver nitrate container, and remove it at least six feet from the working area.
- Tightly close the lid on the mixing container. Turn the container on its side and slowly roll until the two powders are completely mixed. The mixture is now ready for use.
- A good practice is to keep the silver nitrate crystals and the magnesium powder or filings in separate air-tight containers and mix just before use.
Caution:
This mixture should be kept out of direct sunlight to avoid decomposition of the silver nitrate which could render this igniter mixture ineffective.
d. Application.
- Carefully pour or spoon the mixture, in a single pile, on the incendiary. Prepare the mixture for ignition with either Fuse Cord (0101) or Improvised String Fuse (0102) in the normal manner. The fuse cord should terminate near the center of the igniter mixture. Concentrated Sulfuric Acid (0103) and Water (0104) can be used as initiators but are generally less convenient. Ignition takes place almost immediately on contact with the acid or water. These liquid initiators are convenient for use with specific delay mechanisms found in chapter 5.
- When used to ignite flammable liquids, wrap a quantity of the mixture in a nonabsorbent material and suspend it inside the container near the open top. The container must remain open for easy ignition and combustion of the flammable liquid.
- To minimize the hazard of premature ignition of flammable liquid vapors, allow at least two feet of fuse length to extend from the top edge of an open container of flammable liquid before lighting the fuse.
a. Description.
- This item consists of white phosphorus dissolved in carbon disulfide. It can be used to ignite the following incendiaries listed in chapter 4: Napalm (0301), Gelled Gasoline (exotic thickeners) (0302), Gelled Gasoline (improvised thickeners) (0303), and Paraffin-Sawdust (0304). It may be used directly as an incendiary on readily flammable material such as rags, dry paper, dry hay, or in the combustible vapor above liquid fuels.
- Ignition is achieved when the volatile solvent, carbon disulfide, evaporates and the white phosphorus comes in contact with air.
Caution:
Never touch white phosphorus directly or allow any of its solutions to touch the skin. Painful burns which heal very slowly may result. White phosphorus sticks must always be stored completely under water. If any of the phosphorus solution is accidently spilled on the skin, immediately flush the affected area with water; then decontaminate the affected area by dabbing with copper sulfate solution.
b. Material and Equipment.
White phosphorus sticks (sometimes called yellow phosphorus).
Carbon disulfide.
Copper sulfate solution.
Tweezers or tongs.
Two glass containers about 8-ounce capacity with lids or stoppers made of glass, earthenware, or metal. Do not use a rubber lid or stopper (carbon disulfide will attack rubber).
c. Preparation.
- Prepare some copper sulfate solution by adding one spoonful of copper sulfate crystals to one of the glass containers. Fill the container with water, place the stopper in the open mouth of the bottle and shake until the crystals dissolve.
- Pour carbon disulfide into the other glass container to one-quarter container volume.
Caution:
Carbon disulfide fumes are poisonous. Always cap an open container of carbon disulfide as soon as possible. Work in a well ventilated area. - With a pair of tweezers remove some sticks of white phosphorus from their storage container. Totally submerge them immediately in the carbon disulfide to bring the level up to one-half full. Be sure that all the phosphorus left in the original container is completely submerged in water before putting the container away. Wash the tweezers immediately in the copper sulfate solution.
- Securely stopper the bottle containing the white phosphorus and carbon disulfide and allow to stand until the white phosphorus dissolves. This usually takes about eight hours. The time required to dissolve white phosphorus can be reduced by shaking the bottle. Be sure that the bottle top does not come off.
- Do not store in direct sunlight because the solution will become ineffective. This solution should never be stored more than three days.
Note.
If carbon disulfide is not available, benzene (benzol) may be used to dissolve the phosphorus. It requires considerable shaking and overnight soaking to get an appreciable amount of phosphorus dissolved in benzene.
Do not
attempt to use red phosphorus for preparing this igniter because it does not behave like white phosphorus.
d. Application.
- To ignite readily flammable material, pour the white phosphorus solution directly onto the material; it will ignite when the solvent evaporates, exposing the white phosphorus to the air. Once the solution is poured, the empty bottle should be discarded immediately because any solution remaining on the bottle will ignite when the solvent evaporates. Do not cover the soaked flammable material because the carbon disulfide must evaporate for ignition to occur.
- The incendiaries mentioned under
Description
above can be initiated by first impregnating crumpled paper or absorbent paper towels with the white phosphorus solution and placing the impregnated paper on the material to be ignited. - Delay times of the phosphorus solution may be varied by the addition of gasoline or toluene (toluol). Add a small quantity of either solvent to the original white phosphorus solution and test the solution each time until the desired delay time is achieved. Delay times of 20 to 30 minutes may be obtained in this manner.
- Check the delay time under conditions expected at the target. Air currents hasten the evaporation of the solvent and decrease delay time. A high ambient temperature will also decrease delay time whereas a low ambient temperature will increase the delay time. This igniter is not reliable at or below freezing temperatures (32° F.)
- To make incendiary paper, soak strips of ordinary writing paper in the phosphoruscarbon disulfide for a few minutes. Remove the paper with a pair of tweezers or tongs and place in a vial filled with water. Be sure to wash off the tweezers immediately in copper sulfide solution. Cap the vial and store until ready to use. To use this incendiary paper, remove the strips of paper with a pair of tweezers, and place among the material to be ignited.